Indexing & Abstracting
Full Text
Review ArticleDOI Number : 10.36811/ojrmi.2020.110009Article Views : 22Article Downloads : 16
The coronavirus family neurological effects: A literature review
Abdulwahab Alahmari
Radiology Specialist, Radiology Department, Saudi Arabia
*Corresponding Author: Abdulwahab Alahmari, Radiology Specialist, Radiology Department, Al-Namas General Hospital, Ministry of Health, Al-Namas City, Saudi Arabia, Tel: +966562428716; Email: afaa99@hotmail.co.uk
Article Information
Aritcle Type: Review Article
Citation: Abdulwahab Alahmari. 2020. The coronavirus family neurological effects: A literature review. O J Radio Med Img. 3: 29-37.
Copyright: This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Copyright © 2020; Abdulwahab Alahmari MRI finding in the olfactory bulb 14, but a temporal epidermoid cyst was found in the scan see (Figure 5).
Publication history:
Received date: 07 August, 2020Accepted date: 17 August, 2020
Published date: 18 August, 2020
Abstract
All the lethal coronaviruses like the middle east respiratory syndrome, severe acute respiratory syndrome coronavirus, or the new coronavirus have showed similar and non-similar effects on the central nerves system. The aim of this paper is to summarize the published findings as neurological effects of the most dangerous three viruses in the coronavirus family and to find the common neuropathological patterns among the three viruses.
Keywords: Brain; MERS-CoV; SARS-CoV; SARS-CoV-2; Neurological effect; Coronavirus
Literature review
The aim of this literature review is to study the neurological effects caused by the middle east respiratory syndrome (MERS-CoV), severe acute respiratory syndrome coronavirus (SARS-CoV), and the new coronavirus known as COVID-19 or (SARS-CoV-2). As well, Finding the common and uncommon patterns of neurological effects among the three of most lethal viruses in the coronavirus family is the focus of this paper.
Methodology
“MERS-CoV, SARS-CoV, SARS-CoV-2” combined with the term “neurological effect” was done to find the relevant papers. The search with the previous terms started in April 5th 2020 and ended in April 16th 2020. Any paper published in a peer reviewed journal from 16th November 2002 (since SARS-CoV started) to 16th April 2020(this paper deadline) were collected. Any paper has no radiological or neurological contribution was excluded see (Figure 1).
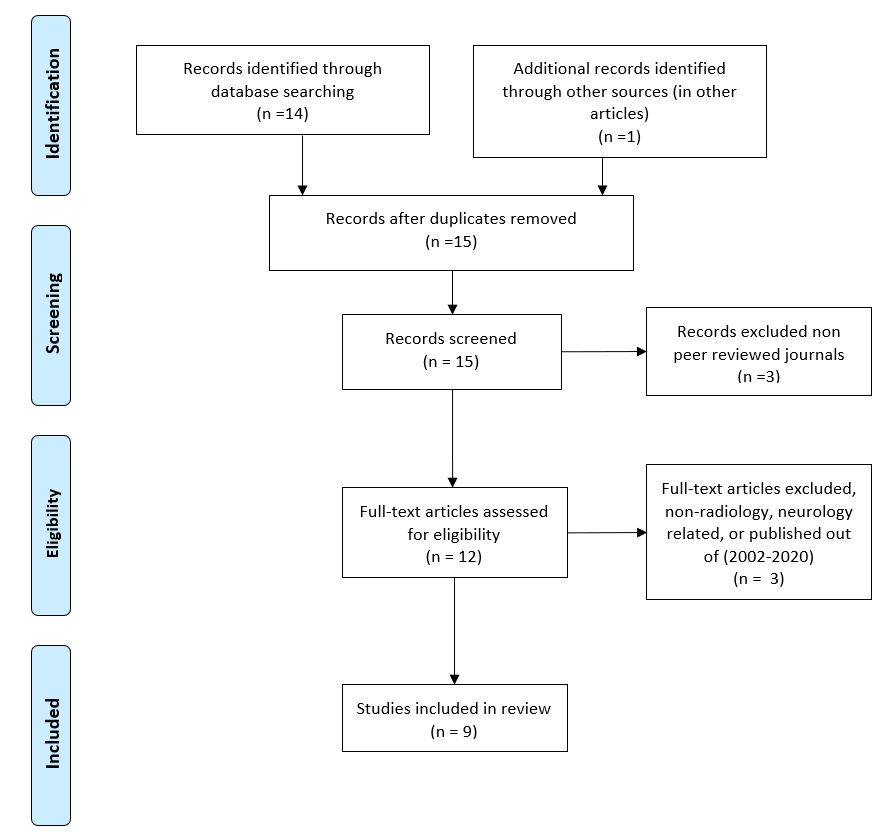
Figure 1: A PRISMA diagram for the selection process.
Introduction
The most lethal viruses in the coronavirus family (i.e. MERS-CoV, SARS-CoV, SARS-CoV-2) showed different effects on the nervous system ranging from mild to life-threatening conditions. The following is an overview of these neurological effects:
Medullary cardiopulmonary centers in the brainstem
Probably the most difficult symptom on the coronavirus patient is not the fever, but it’s the short breath (respiratory distress). Some of the patients required ventilators, so they can breathe. The lethal coronaviruses are usually associated with pneumonia which makes the breathing very difficult. But it has been proved that SARS-CoV passes through chemoreceptors and mechanoreceptors of the lungs from one neuron to another neuron to attack the medullary cardiopulmonary centers in the brainstem through synaptic connections [1]. The breathing becomes more difficult due to the infection in the brainstem. Similarly, SARS-CoV-2 could be transmitted to the brainstem through synaptic connections similar to SARS-CoV [1].
Intracranial hemorrhage
MERS-CoV can cause platelet dysfunction, thrombocytopenia, and disseminated intravenous coagulation which leads to prolonged coagulation profile then will cause a multi-organ failure then irreversible brainstem damage which eventually will lead to death. Polyneuropathy has been reported as non-direct consequences of the MERS-CoV [2]. The brain damage in SARS-CoV case due to hyper-dense lesions has been proven see (Figure 2). There is a SARS-CoV-2 case with thalamic hemorrhage which is a common finding in among the lethal viruses of the coronavirus family see (Figure 2).
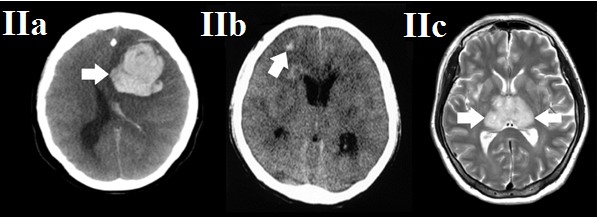
Figure 2: Three medical imaging study of three patients. The patient (A) is 34-year-old female patient with brain hemorrhage (a confirmed case of MERS-COV disease). The patient (B) is 50-year-old female patient with diffuse brain edema and multiple hyper densities lesions suggestive of ischemic and necrotic changes (a confirmed case of SARS-COV disease). The patient (C) is 50 year old female patient with bilateral thalamic hemorrhage (a confirmed case of SARS-COV-2 disease). Findings: The patient (A) study is axial CT scan for the brain show left intracranial hemorrhage, inter-ventricular extension, midline shift, and brain edema (white arrow). The patient (B) study is axial CT scan show multiple hyper-densities -in different slices located upper and lower than the showed slice, but has the same features of the presented scan- with diffuse brain edema (white arrow). The patient (C) study is axial T2 MRI scan show bilateral thalamic hemorrhage that was confirmed as hypo-intense susceptibility weighted images (white arrow). Technique: (A) axial CT, cerebrum window. (B) axial CT, cerebrum window. (C) axial MRI, T2.
Long term neurological sequelae
It has been proven that SARS-CoV mostly enter the brain through the olfactory bulb leading to neurons death. The infection was detected in the brain mostly attacking the cardiopulmonary centers in the brainstem, while a minimal infection presented in the lungs [3].
Extent of the infection in the brain parts
MERS-CoV affect the corticospinal tract, ventricles (causing inter-ventricular hemorrhage), subcortical, basal ganglia, corpus callosum, temporal lobe, parietal lobe, and frontal lobe by different lesions. As well, SARS-CoV affect the olfactory bulb, cerebrum, and cardiopulmonary centers in the brainstem. Similarly, SARS-CoV-2affect the brainstem, thalamus, olfactory bulb, and cerebrum, while the cerebellum remains as the uninfected part of the brain by SARS-CoV-2 [3].
Acute necrotizing encephalopathy (cytokine storm syndrome)
There isa confirmed case of SARS-CoV-2 of a women with bilateral thalamic and temporal lobe damage on both CT and MRI scans [4]. The findings are bilateral medial temporal lobe and thalamic bleedings on FLAIR and they were confirmed by susceptibility weighted images [4]. These findings are similar to another published case of influenza A virus subtype H3 with a fatal acute necrotic encephalitis [5]. The disruption of blood-brain barrier is associated with cytokine release which causes the bleeding.
Symptoms & neurological presentations in coronavirus family
The common patient presentations of MERS-CoV are; low mental functions, headache, seizures, dizziness, limb numbness, and nausea [2]. The common patient presentations of SARS-CoV are; low mental functions, headache, and vomiting 3. The common patient presentations of SARS-CoV-2 are; low mental functions, headache, vomiting, and anosmia & ageusia (olfactory and taste disorders (OTDs)) [4,6].
Hyperthermia vs. brain
The normal body temperature is 37.7 ?C (99.9 ?F) and any temperature beyond 40 ?C can be life-threatening temperature. The developing brain is at risk form hyperthermia. As well, the developed brain can have some effects which depends on the degree of the temperature and the duration of the exposure. In case of a severe hyperthermia, it could induce neuronal necrotic death. Even with lower temperatures, hyperthermia-induced damage can cause neurons to show a late apoptotic characteristic. The mechanism of hyperthermia-induced damage is done by depolarizing the mitochondria in the neurons which would lead to cell damage [7].
Low oxygen saturation vs. brain
The normal oxygen level in the blood is 75 to 100 mm HG and if the oxygen level dropped below 60 mm HG, the patient will need a ventilator. The decrease in oxygen level in the blood will lead to a hypoxia. Brain hypoxia can be categorizing into 4 categories; diffuse cerebral hypoxia, focal cerebral ischemia, massive cerebral infarction, and global cerebral hypoxia. Similar to the body temperature, the low oxygen saturation effect on the nervous system has two important factors; the amount of oxygen in the blood and the period of shortage. The categories of hypoxia are associated with the previously mentioned two factors. The world health organization report claim that the mean incubation time for SARS-CoV-2 is 5.2 days based on a study among 425 patients in Wuhan and it may widely vary among patients which depends on their immune systems [8]. Another paper claims that the SARS-CoV-2 incubation period is 6.4 days [9]. If a patient have a low oxygen saturation for a long time, it could lead to white matter periventricular lesions, infarctions, or cognitive impairments. It is similar to chronic obstructive pulmonary diseases (COPD). The COPD patients with low oxygen saturation, they could develop white matter periventricular and subcortical lesions which will lead to cognitive impairments and mental illness [10].
Meningitis and encephalitis
One SARS-CoV-2 case was reported to be associated with Meningitis and encephalitis [11]. In the report, the patient brought to the hospital due to convulsion and loss of conciseness not because of fever or respiratory issues. The patient’s MRI scan revealed ventricular hyper-intensities on DWI, partially atrophied hippocampus, high signal intensity in the temporal lobe and hippocampus on FLAIR, and pan sinusitis on T2 [11].
White matter lesions
MERS-CoV can causes lesions in the white matter in different regions like; subcortical, basal ganglia, corpus callosum, temporal lobe, parietal lobe, and frontal lobe see (Figure 3). The white matter lesions will cause ataxia, focal motor deficit, and coma [12]. All the brain lesions in the MERS-CoV patient did not show any gadolinium enhancement on MRI [12].

Figure 3: Both MRI scans on top (3a and 3b) for a 74-year-old male patient show multiple bilateral subcortical white matter lesions. The MRI scans (3c, 3d, and 3f) for a 57-year-old male patient show bilateral subcortical white lesions. The MRI scan (3g) for a 45-year-old male patient shows corticospinal tract abnormality (all are confirmed MERS-CoV). Findings: The FLAIR image (3a) shows multiple hyper-intense lesions in the corpus callosum and in the deep white matter and subcortical lesions bilaterally in the frontal, temporal, parietal lobes (white arrow). The DWI image (3b) shows multiple white matter lesions (white arrow). The DWI and ADC images (3c and 3d) show diffusion restriction of the corpus callosum and bilaterally in the frontal lobe (black and white stars respectively). The T2 image (3f) shows bilateral hyper-intense white matter lesions in the corpus callosum and frontal lobe (white arrow). The T2 weighted image (3g) shows abnormality affecting the corticospinal tract bilaterally (white arrow). Technique: (3a) axial MRI, FLAIR image. (3b and 3c) axial MRI, DWI image. (3d) axial MRI, ADC image. (3f) coronal MRI, T2 image. (3g) coronal MRI, T2 weighted image.
Olfactory nerve
It has been proven that the olfactory nerve is affected by SARS-CoV-2 in acute stages with no nasal congestion [13]. In such cases, the MRI images will show a high signal intensity in the olfactory bulb see (Figure 4). As well, anosmia and olfactory neuropathy was reported in SARS-CoV with no abnormal MRI finding in the olfactory bulb [14], but a temporal epidermoid cyst was found in the scan see (Figure 5).
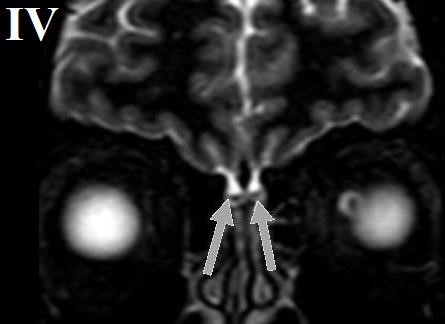
Figure 4: A 27 year−old−male patient with ansomnia and olfactory bulb neuropathy (a SARS-CoV-2 confirmed case). Findings: A T2 wighted image, coronal slice, MRI scan shows a normal volume, no nasal congestion, and olfactory bulb signal intensity (grey arrows). Technique: Coronal MRI, T2 wieghted image.
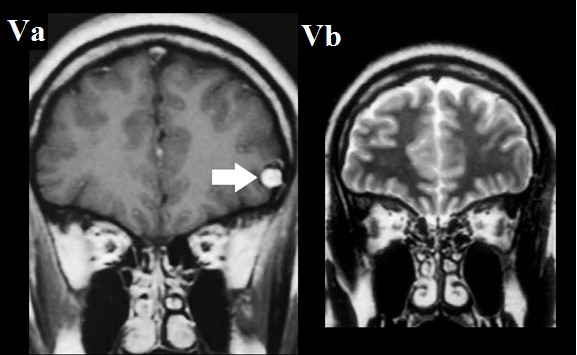
Figure 5: A 27−year−old famale patient with ansomnia and temporal epidermoid cyst. Findings:
A coronal T1 MRI image (5a) shows a tempral epidermoid cyston the left side. A coronal T2 MRI image (5b) shows no abnormal findings in the olfactory bulb (white arrow). Technique: (5a) coronal MRI, T1 wighted image. (5b) coronal MRI, T2 wighted image.
Lack of post-mortem investigation
There is a reporting shortage in the literature in post-mortem cases by medical imaging and autopsy for the coronavirus cases due to infection control reasons. This lack causes a huge misunderstanding of these viruses and very minimal knowledge of their mechanisms in the central nervous system [1]. As well, due to the focus on the respiratory system, there is a neglect and lack of papers that helps in documenting thevarious effects that is caused by the lethal coronaviruses on the nervous system. Even though, SARS-CoV and MARS-CoV started in 2002 and in 2012 respectively, but still there is a lack of information see (Table 1).
|
table 1: The published papers about neurological effect of thecoronavirus family supported by radiology scans. |
||||
|
Author |
Publishing Year |
Journal |
Article Type |
Neurological Findings |
|
Li YC et al. |
2019 |
Journal of medical virology |
Review |
Medullary cardiopulmonary centers affected by SARS-CoV-2 which cause death. |
|
Algahtani H, et al. |
2016 |
Case reports in neurological medicine |
Case report and review of the literature |
MERS-CoV cause brainstem irreversible damage then death. |
|
Netland J, et al. |
2008 |
Journal of virology |
Animal experimental study |
SARS-CoV enter the brain through the olfactory nerve then attack the cardiopulmonary centers in the midbrain. |
|
Poyiadji N, et al. |
2020 |
Radiology |
Case report |
SARS-CoV-2 cause acute necrotizing encephalopathy |
|
Hjelmesæth J, et al. |
2020 |
Tidsskrift for Den norske legeforening |
Case report |
SARS-CoV-2 cause loss of smell and taste |
|
Moriguchi T, et al. |
2020 |
International Journal of Infectious Diseases |
Case report |
SARS-CoV-2 cause Meningitis/Encephalitis |
|
Arabi YM, et al. |
2015 |
Infection |
Case report |
MERS-CoV affected the white matter |
|
Galougahi MK, et al. |
2020 |
Academic radiology |
Case report |
Confirm olfactory bulb neuropathy in SARS-CoV-2 on MRI. |
|
Hwang C. |
2006 |
Acta Neurologica Taiwanica |
Case report |
Can’t confirm the olfactory bulb neuropathy on MRI in SARS-CoV with temporal lobe findings. |
Conclusion
Coronaviruses have a huge effect that is targeting the central nervous system which leads to other symptoms like; vomiting, anosmia, ageusia, respiratory distress, etc. The real issue is happening in the brainstem more than the lungs or the gastrointestinal system which is primarily led to death in some vulnerable cases with chronic conditions. Medical imaging investigation of the central nervous system must be done like the chest CT or swab test for confirmed coronavirus cases. To tackle the issue, an MRI must be done for the confirmed cases to detect any abnormality in the brain and especially the brainstem of the corona patients. A post-mortem pan CT can help in identifying other affected systems and our understanding of these viruses' mechanisms in damaging the human body. The etiology of coronavirus family summarized see (Table 2).
|
Table 2: Etiology table. |
||
|
Lethal Coronavirus Type |
Confirmed Etiologies |
Radiological Patterns |
|
MERS-CoV |
Intracranial hemorrhage |
Intracranial hemorrhage, inter−ventricular extension, midline shift, andbrain edema. |
|
Brainstem damage
|
High signal corticospinal tract extending above the brainstem. |
|
|
White matter lesions |
Affect the subcortical, basal ganglia, corpus callosum, temporal lobe, parietal lobe, and frontal lobe. No gadolinium enhancement. |
|
|
SARS-CoV |
Intracranial hemorrhage |
Multiple hyper−densities and diffuse brain edema. |
|
Olfactory neuropathy |
N/A. |
|
|
SARS-CoV-2 |
Intracranial hemorrhage |
Bilateral thalamic and temporal lobe hemorrhages. |
|
Olfactory neuropathy
|
High signal olfactory bulb on T2 wighted images. Normal nasal volume (no congestion). |
|
|
Meningitis and encephalitis
|
Ventricular hyper-intensities on DWI . Partially atrophied structure like the hippocampus on FLAIR. High signal temporal lobe and hippocampus on FLAIR. Pan sinusitis on T2. |
|
|
Acute necrotizing encephalopathy (cytokine storm syndrome) |
Bilateral medial temporal lobes and bilateral thalamic bleeding on FLAIR confirmed by susceptibility weighted images. |
|
Teaching points
There is a large similarity between the lethal viruses in the coronavirus family especially in their effects on the nervous system. The central nervous system is largely involved in the coronavirus infection and we must pay more attention to this system which could help in saving lives.
Abbreviations
MERS-CoV: Middle east respiratory syndrome.
SARS-CoV: Severe acute respiratory syndrome coronavirus.
SARS-CoV-2: Severe acute respiratory syndrome coronavirus-2.
OTDs: Olfactory and taste disorders.
COPD: Chronic obstructive pulmonary diseases.
MRI: Magnetic resonance imaging.
CT: Computed tomography.
FLAIR: Fluid attenuated inversion recovery.
DWI: Diffusion weighted imaging.
ADC: Apparent diffusion coefficient.
T1: Time 1.
T2: Time 2.
mm HG: Millimeter of mercury (pressure unit).
?C: Celsius or centigrade scale (temperature unit).
?F: Fahrenheit scale (temperature unit).
References
1. Li YC, Bai WZ, Hashikawa T. 2020. The neuroinvasive potential of SARS?CoV2 may be at least partially responsible for the respiratory failure of COVID?19 patients. Journal of medical virology. Ref.: shorturl.at/jvzU8
2. Algahtani H, Subahi A, Shirah B. 2016. Neurological complications of Middle East respiratory syndrome coronavirus: a report of two cases and review of the literature. Case reports in neurological medicine. Ref.: shorturl.at/aizV3
3. Netland J, Meyerholz DK, Moore S, et al. 2008. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. Journal of virology. 82: 7264-7275. Ref.: shorturl.at/mwVX8
4. Poyiadji N, Shahin G, Noujaim D, et al. 2020. COVID-19-associated Acute Hemorrhagic Necrotizing Encephalopathy: CT and MRI Features. Radiology. 31: 201187. Ref.: shorturl.at/CEM27
5. Pulakanti V, Holland N. 2012. A Fatal Case of Adult-Onset Acute Necrotizing Encephalitis Secondary to Influenza A Virus. Neurology. Apr (P5. 398). Ref.: shorturl.at/inIKM
6. Hjelmesæth J, Skaare D. 2020. Loss of smell or taste as the only symptom of COVID-19. Tidsskrift for Den norske legeforening. Ref.: shorturl.at/huJ12
7. White MG, Luca LE, Nonner D, et al. 2007. Cellular mechanisms of neuronal damage from hyperthermia. Progress in brain research. 162: 347-371. Ref.: shorturl.at/ivLV3
8. World Health Organization. 2020. Laboratory testing for coronavirus disease 2019 (COVID-19) in suspected human cases: interim guidance, 2 March 2020. World Health Organization. Ref.: shorturl.at/hxG35
9. Jiang X, Rayner S, Luo MH. 2020. Does SARS?CoV?2 has a longer incubation period than SARS and MERS?. Journal of medical virology. Feb 24. Ref.: shorturl.at/imrDS
10. Areza-Fegyveres R, Kairalla RA, Carvalho CR, et al. 2010. Cognition and chronic hypoxia in pulmonary diseases. Dementia & neuropsychologia. 4: 14-22. Ref.: shorturl.at/nsBMO
11. Moriguchi T, Harii N, Goto J, et al. 2020. A first Case of Meningitis/Encephalitis associated with SARS-Coronavirus-2. International Journal of Infectious Diseases. Ref.: shorturl.at/asCQU
12. Galougahi MK, Ghorbani J, Bakhshayeshkaram M, et al. Olfactory bulb magnetic resonance imaging in SARS-CoV-2-induced anosmia: The first report. Academic Radiology. Ref.: shorturl.at/wBCG0
13. Hwang C. 2006. Olfactory Neuropathy in Severe Acute Respiratory Syndrome: Report of A Case. Acta Neurologica Taiwanica. 15: 26. Ref.: shorturl.at/cknu2




















